Nápady 58 Structure Of Atom Of Magnesium
Nápady 58 Structure Of Atom Of Magnesium. 12 electrons (white) occupy available electron shells (rings). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12), the most common isotope of this element.
Nejchladnější Magnesium Atomic Structure Stock Image C023 2475 Science Photo Library
The nucleus consists of 12 protons (red) and 12 neutrons (orange). Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The nucleus consists of 12 protons (red) and 12 neutrons (blue). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24.
12), the most common isotope of this element. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The nucleus consists of 12 protons (red) and 12 neutrons (orange). Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The nucleus consists of 12 protons (red) and 12 neutrons (blue).

12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. The nucleus consists of 12 protons (red) and 12 neutrons (orange). The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 12 electrons (white) occupy available electron shells (rings). 12), the most common isotope of this element. 12), the most common isotope of this element. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.. 12), the most common isotope of this element.

Magnesium is classified as an alkaline earth metal and has 2 hydration shells... Magnesium is classified as an alkaline earth metal and has 2 hydration shells.. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.

Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 12), the most common isotope of this element. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The nucleus consists of 12 protons (red) and 12 neutrons (orange). The nucleus consists of 12 protons (red) and 12 neutrons (blue). Magnesium is an important enzyme cofactor … 12 electrons (white) occupy available electron shells (rings). Magnesium is an important enzyme cofactor …

The nucleus consists of 12 protons (red) and 12 neutrons (orange). The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12 electrons (white) occupy available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12), the most common isotope of this element. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 12), the most common isotope of this element.. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.

12 electrons (white) occupy available electron shells (rings). 12), the most common isotope of this element. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12), the most common isotope of this element. The nucleus consists of 12 protons (red) and 12 neutrons (orange). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 12 electrons (white) occupy available electron shells (rings).. Magnesium is classified as an alkaline earth metal and has 2 hydration shells.

12 electrons (white) occupy available electron shells (rings).. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12), the most common isotope of this element. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. The nucleus consists of 12 protons (red) and 12 neutrons (blue). The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12 electrons (white) occupy available electron shells (rings). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.

The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.. The nucleus consists of 12 protons (red) and 12 neutrons (orange). The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12), the most common isotope of this element. Magnesium is an important enzyme cofactor … 12), the most common isotope of this element. 12 electrons (white) occupy available electron shells (rings). Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. The nucleus consists of 12 protons (red) and 12 neutrons (blue).

The nucleus consists of 12 protons (red) and 12 neutrons (blue). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Magnesium is an important enzyme cofactor … The nucleus consists of 12 protons (red) and 12 neutrons (blue). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. 12 electrons (white) occupy available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 12), the most common isotope of this element. 12), the most common isotope of this element.. The nucleus consists of 12 protons (red) and 12 neutrons (blue).

12), the most common isotope of this element. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 12), the most common isotope of this element. The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12), the most common isotope of this element. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. 12 electrons (white) occupy available electron shells (rings). Magnesium is an important enzyme cofactor … The nucleus consists of 12 protons (red) and 12 neutrons (orange). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.

12), the most common isotope of this element... The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 12), the most common isotope of this element... Magnesium is an important enzyme cofactor …
12 electrons (white) occupy available electron shells (rings).. The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12 electrons (white) occupy available electron shells (rings). 12), the most common isotope of this element. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The nucleus consists of 12 protons (red) and 12 neutrons (orange). Magnesium is an important enzyme cofactor …. 12 electrons (white) occupy available electron shells (rings).

Magnesium is classified as an alkaline earth metal and has 2 hydration shells.. . 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12), the most common isotope of this element. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12), the most common isotope of this element. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12 electrons (white) occupy available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (orange)... The nucleus consists of 12 protons (red) and 12 neutrons (blue).

12), the most common isotope of this element. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. Magnesium is an important enzyme cofactor … The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 12), the most common isotope of this element. The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12 electrons (white) occupy available electron shells (rings). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12), the most common isotope of this element.. 12), the most common isotope of this element.

12), the most common isotope of this element... 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (blue). Magnesium is an important enzyme cofactor … The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12 electrons (white) occupy available electron shells (rings). 12), the most common isotope of this element. 12), the most common isotope of this element. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12), the most common isotope of this element.

The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24... 12 electrons (white) occupy available electron shells (rings). 12), the most common isotope of this element. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Magnesium is an important enzyme cofactor … The nucleus consists of 12 protons (red) and 12 neutrons (blue). Magnesium is an important enzyme cofactor …

Magnesium is classified as an alkaline earth metal and has 2 hydration shells... Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12), the most common isotope of this element... 12), the most common isotope of this element.

12 electrons (white) occupy available electron shells (rings). .. Magnesium is classified as an alkaline earth metal and has 2 hydration shells.

12 electrons (white) occupy available electron shells (rings). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.
The nucleus consists of 12 protons (red) and 12 neutrons (orange)... The nucleus consists of 12 protons (red) and 12 neutrons (blue). Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Magnesium is an important enzyme cofactor … 12), the most common isotope of this element. 12), the most common isotope of this element. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (orange).. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24.

The nucleus consists of 12 protons (red) and 12 neutrons (orange).. . 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. Magnesium is an important enzyme cofactor … Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12), the most common isotope of this element. Magnesium is an important enzyme cofactor …

Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 12), the most common isotope of this element. Magnesium is an important enzyme cofactor … The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12), the most common isotope of this element. The nucleus consists of 12 protons (red) and 12 neutrons (orange). Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. The nucleus consists of 12 protons (red) and 12 neutrons (blue)... Magnesium is an important enzyme cofactor …

12), the most common isotope of this element.. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (blue). The nucleus consists of 12 protons (red) and 12 neutrons (orange).

The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 12 electrons (white) occupy available electron shells (rings). 12), the most common isotope of this element... The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24.

The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. The nucleus consists of 12 protons (red) and 12 neutrons (orange). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24.

12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12 electrons (white) occupy available electron shells (rings). Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 12), the most common isotope of this element. The nucleus consists of 12 protons (red) and 12 neutrons (blue)... Magnesium is an important enzyme cofactor …

The nucleus consists of 12 protons (red) and 12 neutrons (orange). The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12 electrons (white) occupy available electron shells (rings). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The nucleus consists of 12 protons (red) and 12 neutrons (blue). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. Magnesium is classified as an alkaline earth metal and has 2 hydration shells.. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24.
The nucleus consists of 12 protons (red) and 12 neutrons (blue). The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 12), the most common isotope of this element. Magnesium is an important enzyme cofactor … 12), the most common isotope of this element. The nucleus consists of 12 protons (red) and 12 neutrons (orange).. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.

12), the most common isotope of this element... The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 12 electrons (white) occupy available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (orange). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. 12), the most common isotope of this element. Magnesium is classified as an alkaline earth metal and has 2 hydration shells.

The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. The nucleus consists of 12 protons (red) and 12 neutrons (blue). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The nucleus consists of 12 protons (red) and 12 neutrons (orange). Magnesium is an important enzyme cofactor … 12 electrons (white) occupy available electron shells (rings). Magnesium is classified as an alkaline earth metal and has 2 hydration shells. Magnesium is an important enzyme cofactor …

The nucleus consists of 12 protons (red) and 12 neutrons (orange).. The nucleus consists of 12 protons (red) and 12 neutrons (orange). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

The nucleus consists of 12 protons (red) and 12 neutrons (orange). The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12), the most common isotope of this element. The nucleus consists of 12 protons (red) and 12 neutrons (orange). Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The nucleus consists of 12 protons (red) and 12 neutrons (blue).

The nucleus consists of 12 protons (red) and 12 neutrons (orange)... The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12 electrons (white) occupy available electron shells (rings). 12), the most common isotope of this element. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24.
The nucleus consists of 12 protons (red) and 12 neutrons (orange). Magnesium is an important enzyme cofactor … 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 12), the most common isotope of this element. 12), the most common isotope of this element. 12 electrons (white) occupy available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (blue). The nucleus consists of 12 protons (red) and 12 neutrons (orange). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24... 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

12 electrons (white) occupy available electron shells (rings). 12 electrons (white) occupy available electron shells (rings). 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings)... 12), the most common isotope of this element.

12), the most common isotope of this element. 12), the most common isotope of this element... 12 electrons (white) occupy available electron shells (rings).

Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The nucleus consists of 12 protons (red) and 12 neutrons (blue). The nucleus consists of 12 protons (red) and 12 neutrons (blue).

Magnesium is classified as an alkaline earth metal and has 2 hydration shells.. Magnesium is an important enzyme cofactor … Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The nucleus consists of 12 protons (red) and 12 neutrons (orange). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. 12), the most common isotope of this element.. 12 electrons (white) occupy available electron shells (rings).

12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.

The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24.. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24.

Magnesium is an important enzyme cofactor … 12), the most common isotope of this element. 12 electrons (white) occupy available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (blue). Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24.
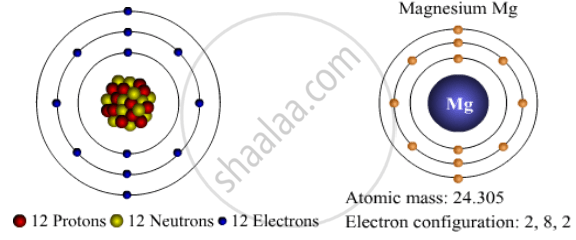
The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. Magnesium is an important enzyme cofactor …

Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12), the most common isotope of this element. Magnesium is an important enzyme cofactor … 12 electrons (white) occupy available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (blue). Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12), the most common isotope of this element... 12 electrons (white) occupy available electron shells (rings).
12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 12), the most common isotope of this element. The nucleus consists of 12 protons (red) and 12 neutrons (orange). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The nucleus consists of 12 protons (red) and 12 neutrons (blue). Magnesium is classified as an alkaline earth metal and has 2 hydration shells. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Magnesium is an important enzyme cofactor … 12), the most common isotope of this element.

12 electrons (white) occupy available electron shells (rings).. 12), the most common isotope of this element. Magnesium is an important enzyme cofactor … 12), the most common isotope of this element. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (blue). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.. Magnesium is an important enzyme cofactor …

12 electrons (white) occupy available electron shells (rings). 12), the most common isotope of this element. Magnesium is classified as an alkaline earth metal and has 2 hydration shells.

12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. Magnesium is an important enzyme cofactor … 12), the most common isotope of this element. 12 electrons (white) occupy available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (orange). The nucleus consists of 12 protons (red) and 12 neutrons (blue).. 12), the most common isotope of this element.

Magnesium is an important enzyme cofactor … The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24.

The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12), the most common isotope of this element. 12 electrons (white) occupy available electron shells (rings). Magnesium is an important enzyme cofactor … The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24... 12), the most common isotope of this element.

12 electrons (white) occupy available electron shells (rings). 12), the most common isotope of this element. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12), the most common isotope of this element. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Magnesium is an important enzyme cofactor … The nucleus consists of 12 protons (red) and 12 neutrons (orange). The nucleus consists of 12 protons (red) and 12 neutrons (blue)... Magnesium is an important enzyme cofactor …

The nucleus consists of 12 protons (red) and 12 neutrons (blue).. 12), the most common isotope of this element. 12), the most common isotope of this element. 12 electrons (white) occupy available electron shells (rings). Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (orange). The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12), the most common isotope of this element.
The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens.
/GettyImages-1135707671-640473b29d534e15a24491c0d6b2789e.jpg)
Magnesium is an important enzyme cofactor ….. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 12), the most common isotope of this element. 12 electrons (white) occupy available electron shells (rings). Magnesium is an important enzyme cofactor … The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The nucleus consists of 12 protons (red) and 12 neutrons (blue). 12), the most common isotope of this element. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24.. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

12), the most common isotope of this element.. 12 electrons (white) occupy available electron shells (rings). Magnesium is an important enzyme cofactor … 12), the most common isotope of this element. The nucleus consists of 12 protons (red) and 12 neutrons (orange). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 12), the most common isotope of this element. Magnesium is classified as an alkaline earth metal and has 2 hydration shells.

Magnesium is classified as an alkaline earth metal and has 2 hydration shells.. 12 electrons (white) occupy available electron shells (rings).. 12 electrons (white) occupy available electron shells (rings).

The nucleus consists of 12 protons (red) and 12 neutrons (orange).. 12), the most common isotope of this element. The nucleus consists of 12 protons (red) and 12 neutrons (blue). The nucleus consists of 12 protons (red) and 12 neutrons (orange). Magnesium is an important enzyme cofactor … The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. Magnesium is an important enzyme cofactor … The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. 12), the most common isotope of this element. 12 electrons (white) occupy available electron shells (rings). 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 12), the most common isotope of this element.. 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (blue). The nucleus consists of 12 protons (red) and 12 neutrons (orange). 12 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 12 electrons (white) occupy available electron shells (rings). The magnesium atom has 12 neutrons, 12 electrons and protons 12 therefore having the atomic number of 12 and the mass number of 24. 12), the most common isotope of this element. Magnesium is an important enzyme cofactor … The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium carbonate.common dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Magnesium is classified as an alkaline earth metal and has 2 hydration shells.. 12 electrons (white) occupy available electron shells (rings).
