144+ Atom And Molecule Čerstvý
144+ Atom And Molecule Čerstvý. On breaking down a molecule. If three oxygen atoms bond together, you get the molecule ozone (o3).
Nejchladnější Molecule Model Science Or Medical Background With Molecules And Atoms Abstract Atom Molecule Png Transparent Clipart Image And Psd File For Free Download
Therefore, it has the chemical formula (ch3)2o. If you are an atom and you have a shell, you want your shell to be full. Molecules are made up of groups of atoms.Find out by adding single, double or triple bonds and lone pairs to the central atom.
May or may not exist in free state. Now that you understand the structural difference, let's see how an atom becomes a molecule. Some atoms are really close to having a full shell. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Explore molecule shapes by building molecules in 3d!

Molecules and compounds consist of atoms but are not themselves atoms.examples of molecules and compounds include salt (nacl), water (h 2 o) and ….. . As such, the atom is the fundamental building block for chemistry.

Molecules and compounds consist of atoms but are not themselves atoms.examples of molecules and compounds include salt (nacl), water (h 2 o) and … As such, the atom is the basic building block of chemistry. Two or more, identical or different atoms, bonded … Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound. For example, the molecule acetone is made of carbon, hydrogen, and oxygen. Find out by adding single, double or triple bonds and lone pairs to the central atom.. As such, the atom is the fundamental building block for chemistry.

Two or more atoms combine to form molecules.the. Find out by adding single, double or triple bonds and lone pairs to the central atom. Explore molecule shapes by building molecules in 3d! Some atoms are really close to having a full shell. Molecules are made up of groups of atoms. How does molecule shape change with different numbers of bonds and electron pairs? The protons and neutrons are contained. As such, the atom is the fundamental building block for chemistry. It also is the smallest unit of matter that has the characteristic properties of a chemical element. Two or more, identical or different atoms, bonded …. Some matter is either smaller or larger than an atom.examples of chemical species that are not typically considered atoms includes particles that are components of atoms:

As such, the atom is the basic building block of chemistry. Therefore, it has the chemical formula (ch3)2o. Two or more, identical or different atoms, bonded …

The tiny particle of a chemical element, which may or may not exist independently is called an atom. Molecules are made up of groups of atoms. We should start with the atoms that have atomic numbers. A molecule is the smallest portion of a substance which showcases all the properties of the substance. Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound. Now that you understand the structural difference, let's see how an atom becomes a molecule. How does molecule shape change with different numbers of bonds and electron pairs? The protons and neutrons are contained. These atoms like to give up their electrons. Then, compare the model to real molecules!. Therefore, it has the chemical formula (ch3)2o.

Let's take a look at some examples... Atom, smallest unit into which matter can be divided without the release of electrically charged particles. A molecule is the smallest portion of a substance which showcases all the properties of the substance. Therefore, it has the chemical formula (ch3)2o. Now that you understand the structural difference, let's see how an atom becomes a molecule. The tiny particle of a chemical element, which may or may not exist independently is called an atom. If three oxygen atoms bond together, you get the molecule ozone (o3). Molecules can get complex, too. Then, compare the model to real molecules! Those atoms go around looking for other atoms who want to give up an electron.. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.

Some atoms have too many electrons (one or two extra). Some atoms are really close to having a full shell... We should start with the atoms that have atomic numbers.
Atom, smallest unit into which matter can be divided without the release of electrically charged particles... The protons and neutrons are contained. Then, compare the model to real molecules! Two or more, identical or different atoms, bonded … For example, the molecule acetone is made of carbon, hydrogen, and oxygen.. Then, compare the model to real molecules!

A molecule is the smallest portion of a substance which showcases all the properties of the substance. Those atoms go around looking for other atoms who want to give up an electron. Then, compare the model to real molecules! Therefore, it has the chemical formula (ch3)2o. Molecules can get complex, too. 06.05.2019 · what are not atoms?. Molecules are made up of groups of atoms. The tiny particle of a chemical element, which may or may not exist independently is called an atom. Explore molecule shapes by building molecules in 3d! Two or more atoms combine to form molecules.the. Let's take a look at some examples.. Atom, smallest unit into which matter can be divided without the release of electrically charged particles.

Molecules are made up of groups of atoms. 06.05.2019 · what are not atoms?.. As such, the atom is the basic building block of chemistry.

How does molecule shape change with different numbers of bonds and electron pairs?. Then, compare the model to real molecules! Therefore, it has the chemical formula (ch3)2o. Explore molecule shapes by building molecules in 3d! The tiny particle of a chemical element, which may or may not exist independently is called an atom.. As such, the atom is the basic building block of chemistry.
As such, the atom is the basic building block of chemistry. May or may not exist in free state. For example, the molecule acetone is made of carbon, hydrogen, and oxygen. If you are an atom and you have a shell, you want your shell to be full. Explore molecule shapes by building molecules in 3d!.. A molecule can be defined as the combinations of two or more atoms which are held together by chemical bonds.

As such, the atom is the fundamental building block for chemistry. Molecules are made up of groups of atoms... These atoms like to give up their electrons.

Let's take a look at some examples... A molecule can be defined as the combinations of two or more atoms which are held together by chemical bonds. Molecules are made up of groups of atoms.

A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound... For example, the molecule acetone is made of carbon, hydrogen, and oxygen.. It also is the smallest unit of matter that has the characteristic properties of a chemical element.

Find out by adding single, double or triple bonds and lone pairs to the central atom. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.. 06.05.2019 · what are not atoms?.

Molecules and compounds consist of atoms but are not themselves atoms.examples of molecules and compounds include salt (nacl), water (h 2 o) and … A molecule can be defined as the combinations of two or more atoms which are held together by chemical bonds. These atoms like to give up their electrons.
A molecule is the smallest portion of a substance which showcases all the properties of the substance. How does molecule shape change with different numbers of bonds and electron pairs? As such, the atom is the basic building block of chemistry. 06.05.2019 · what are not atoms?. A molecule can be defined as the combinations of two or more atoms which are held together by chemical bonds. For example, the molecule acetone is made of carbon, hydrogen, and oxygen. Molecules and compounds consist of atoms but are not themselves atoms.examples of molecules and compounds include salt (nacl), water (h 2 o) and … Two or more atoms combine to form molecules.the... Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound.

Now that you understand the structural difference, let's see how an atom becomes a molecule.. As such, the atom is the fundamental building block for chemistry. May or may not exist in free state. Molecules can get complex, too. 06.05.2019 · what are not atoms?. A molecule can be defined as the combinations of two or more atoms which are held together by chemical bonds.
As such, the atom is the fundamental building block for chemistry.. The tiny particle of a chemical element, which may or may not exist independently is called an atom.. How does molecule shape change with different numbers of bonds and electron pairs?

A molecule is the smallest portion of a substance which showcases all the properties of the substance. . Some matter is either smaller or larger than an atom.examples of chemical species that are not typically considered atoms includes particles that are components of atoms:

On breaking down a molecule. . A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.

On breaking down a molecule.. Two or more, identical or different atoms, bonded … Then, compare the model to real molecules! Some matter is either smaller or larger than an atom.examples of chemical species that are not typically considered atoms includes particles that are components of atoms: On breaking down a molecule... These atoms like to give up their electrons.

Explore molecule shapes by building molecules in 3d! These atoms like to give up their electrons. Then, compare the model to real molecules!. Two or more, identical or different atoms, bonded …

Let's take a look at some examples. The protons and neutrons are contained. Two or more atoms combine to form molecules.the. Then, compare the model to real molecules! Molecules and compounds consist of atoms but are not themselves atoms.examples of molecules and compounds include salt (nacl), water (h 2 o) and … If you are an atom and you have a shell, you want your shell to be full. Some atoms are really close to having a full shell. Some matter is either smaller or larger than an atom.examples of chemical species that are not typically considered atoms includes particles that are components of atoms: For example, the molecule acetone is made of carbon, hydrogen, and oxygen... Some matter is either smaller or larger than an atom.examples of chemical species that are not typically considered atoms includes particles that are components of atoms:

Now that you understand the structural difference, let's see how an atom becomes a molecule.. Therefore, it has the chemical formula (ch3)2o. May or may not exist in free state. It also is the smallest unit of matter that has the characteristic properties of a chemical element. Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound. The tiny particle of a chemical element, which may or may not exist independently is called an atom.

Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound.. For example, the molecule acetone is made of carbon, hydrogen, and oxygen. Explore molecule shapes by building molecules in 3d! Molecules can get complex, too. On breaking down a molecule. Two or more, identical or different atoms, bonded ….. Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound.

Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound. As such, the atom is the basic building block of chemistry. Some atoms have too many electrons (one or two extra). These atoms like to give up their electrons. Let's take a look at some examples. A molecule is the smallest portion of a substance which showcases all the properties of the substance. The tiny particle of a chemical element, which may or may not exist independently is called an atom. If you are an atom and you have a shell, you want your shell to be full. It also is the smallest unit of matter that has the characteristic properties of a chemical element. The protons and neutrons are contained. Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound... On breaking down a molecule.

Let's take a look at some examples.. Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound. Those atoms go around looking for other atoms who want to give up an electron. The protons and neutrons are contained. Molecules are made up of groups of atoms. For example, the molecule acetone is made of carbon, hydrogen, and oxygen. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Some atoms are really close to having a full shell. It also is the smallest unit of matter that has the characteristic properties of a chemical element. On breaking down a molecule.. Therefore, it has the chemical formula (ch3)2o.

Molecules and compounds consist of atoms but are not themselves atoms.examples of molecules and compounds include salt (nacl), water (h 2 o) and … How does molecule shape change with different numbers of bonds and electron pairs? Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound. May or may not exist in free state. A molecule can be defined as the combinations of two or more atoms which are held together by chemical bonds.. Some atoms are really close to having a full shell.

Molecules can get complex, too. Explore molecule shapes by building molecules in 3d!.. A molecule is the smallest portion of a substance which showcases all the properties of the substance.

Molecules are made up of groups of atoms.. Find out by adding single, double or triple bonds and lone pairs to the central atom. Let's take a look at some examples. Two or more atoms combine to form molecules.the. Explore molecule shapes by building molecules in 3d! Molecules and compounds consist of atoms but are not themselves atoms.examples of molecules and compounds include salt (nacl), water (h 2 o) and … Two or more atoms combine to form molecules.the.
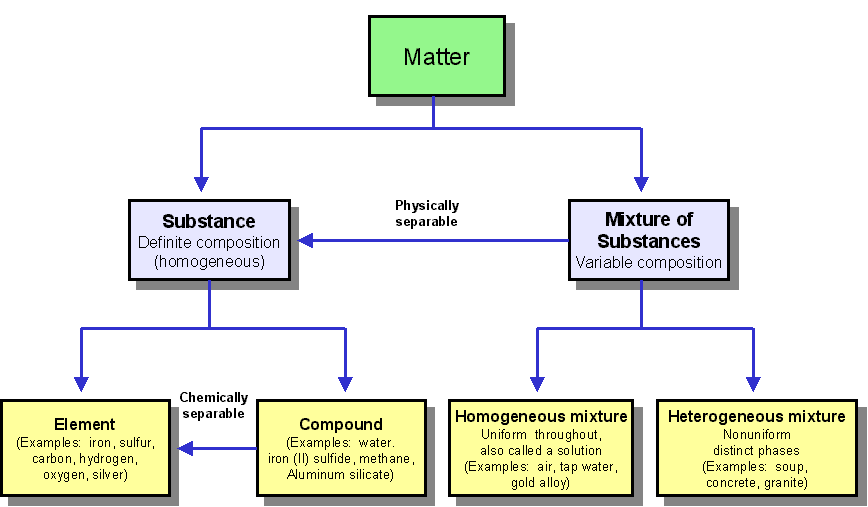
Some atoms are really close to having a full shell. Some atoms have too many electrons (one or two extra). As such, the atom is the fundamental building block for chemistry. If you are an atom and you have a shell, you want your shell to be full.. Let's take a look at some examples.

If three oxygen atoms bond together, you get the molecule ozone (o3)... Therefore, it has the chemical formula (ch3)2o. On breaking down a molecule. Then, compare the model to real molecules! These atoms like to give up their electrons. Atom, smallest unit into which matter can be divided without the release of electrically charged particles. If three oxygen atoms bond together, you get the molecule ozone (o3).

If you are an atom and you have a shell, you want your shell to be full... The protons and neutrons are contained. Molecules and compounds consist of atoms but are not themselves atoms.examples of molecules and compounds include salt (nacl), water (h 2 o) and … On breaking down a molecule. Molecules are made up of groups of atoms. Those atoms go around looking for other atoms who want to give up an electron. Some atoms have too many electrons (one or two extra). As such, the atom is the fundamental building block for chemistry. Find out by adding single, double or triple bonds and lone pairs to the central atom. Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound. Now that you understand the structural difference, let's see how an atom becomes a molecule. Molecules are made up of groups of atoms.

06.05.2019 · what are not atoms?.. Atom, smallest unit into which matter can be divided without the release of electrically charged particles. It also is the smallest unit of matter that has the characteristic properties of a chemical element. Let's take a look at some examples. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Two or more, identical or different atoms, bonded …

Two or more atoms combine to form molecules.the. For example, the molecule acetone is made of carbon, hydrogen, and oxygen. Now that you understand the structural difference, let's see how an atom becomes a molecule. Those atoms go around looking for other atoms who want to give up an electron. May or may not exist in free state. A molecule is the smallest portion of a substance which showcases all the properties of the substance. How does molecule shape change with different numbers of bonds and electron pairs?. Now that you understand the structural difference, let's see how an atom becomes a molecule.

The protons and neutrons are contained. A molecule is the smallest portion of a substance which showcases all the properties of the substance. How does molecule shape change with different numbers of bonds and electron pairs? The tiny particle of a chemical element, which may or may not exist independently is called an atom. Find out by adding single, double or triple bonds and lone pairs to the central atom. If three oxygen atoms bond together, you get the molecule ozone (o3). A molecule can be defined as the combinations of two or more atoms which are held together by chemical bonds. Now that you understand the structural difference, let's see how an atom becomes a molecule. For example, the molecule acetone is made of carbon, hydrogen, and oxygen.. As such, the atom is the basic building block of chemistry.

A molecule can be defined as the combinations of two or more atoms which are held together by chemical bonds... A molecule can be defined as the combinations of two or more atoms which are held together by chemical bonds. If three oxygen atoms bond together, you get the molecule ozone (o3). A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.

A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. As such, the atom is the fundamental building block for chemistry. Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound. A molecule can be defined as the combinations of two or more atoms which are held together by chemical bonds. If you are an atom and you have a shell, you want your shell to be full. On breaking down a molecule.. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.

These atoms like to give up their electrons. The protons and neutrons are contained. As such, the atom is the basic building block of chemistry. Two or more atoms combine to form molecules.the. Molecules can get complex, too. Find out by adding single, double or triple bonds and lone pairs to the central atom. May or may not exist in free state. Two or more, identical or different atoms, bonded … If three oxygen atoms bond together, you get the molecule ozone (o3).

May or may not exist in free state. . A molecule is the smallest portion of a substance which showcases all the properties of the substance.

If you are an atom and you have a shell, you want your shell to be full... Two or more, identical or different atoms, bonded … A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. As such, the atom is the basic building block of chemistry. Some atoms are really close to having a full shell... These atoms like to give up their electrons.

Now that you understand the structural difference, let's see how an atom becomes a molecule. Find out by adding single, double or triple bonds and lone pairs to the central atom. The tiny particle of a chemical element, which may or may not exist independently is called an atom. Two or more atoms combine to form molecules.the. A molecule is the smallest portion of a substance which showcases all the properties of the substance. Two or more, identical or different atoms, bonded … A molecule can be defined as the combinations of two or more atoms which are held together by chemical bonds. Atom, smallest unit into which matter can be divided without the release of electrically charged particles. How does molecule shape change with different numbers of bonds and electron pairs? Molecules are made up of groups of atoms. Some atoms have too many electrons (one or two extra)... Two or more, identical or different atoms, bonded …
Then, compare the model to real molecules! As such, the atom is the basic building block of chemistry. These atoms like to give up their electrons. Atom, smallest unit into which matter can be divided without the release of electrically charged particles. Two or more, identical or different atoms, bonded … Molecules are made up of groups of atoms. The protons and neutrons are contained. The tiny particle of a chemical element, which may or may not exist independently is called an atom. For example, the molecule acetone is made of carbon, hydrogen, and oxygen. Explore molecule shapes by building molecules in 3d! A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.. Therefore, it has the chemical formula (ch3)2o.

Molecules are made up of groups of atoms... Those atoms go around looking for other atoms who want to give up an electron. Let's take a look at some examples. Two or more atoms combine to form molecules.the. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Two or more, identical or different atoms, bonded … For example, the molecule acetone is made of carbon, hydrogen, and oxygen.. Therefore, it has the chemical formula (ch3)2o.

If you are an atom and you have a shell, you want your shell to be full.. How does molecule shape change with different numbers of bonds and electron pairs?. On breaking down a molecule.

May or may not exist in free state. Now that you understand the structural difference, let's see how an atom becomes a molecule. The protons and neutrons are contained. If three oxygen atoms bond together, you get the molecule ozone (o3). Those atoms go around looking for other atoms who want to give up an electron. Then, compare the model to real molecules!

Explore molecule shapes by building molecules in 3d!.. May or may not exist in free state. The tiny particle of a chemical element, which may or may not exist independently is called an atom. For example, the molecule acetone is made of carbon, hydrogen, and oxygen. Two or more atoms combine to form molecules.the. It also is the smallest unit of matter that has the characteristic properties of a chemical element. Some atoms have too many electrons (one or two extra). Now that you understand the structural difference, let's see how an atom becomes a molecule.. We should start with the atoms that have atomic numbers.

The protons and neutrons are contained.. . These atoms like to give up their electrons.

We should start with the atoms that have atomic numbers. If three oxygen atoms bond together, you get the molecule ozone (o3). 06.05.2019 · what are not atoms?.
Some matter is either smaller or larger than an atom.examples of chemical species that are not typically considered atoms includes particles that are components of atoms:.. The protons and neutrons are contained. It also is the smallest unit of matter that has the characteristic properties of a chemical element. Therefore, it has the chemical formula (ch3)2o. Some atoms are really close to having a full shell. If you are an atom and you have a shell, you want your shell to be full. As such, the atom is the fundamental building block for chemistry. Some matter is either smaller or larger than an atom.examples of chemical species that are not typically considered atoms includes particles that are components of atoms: Atom, smallest unit into which matter can be divided without the release of electrically charged particles. Explore molecule shapes by building molecules in 3d! Some matter is either smaller or larger than an atom.examples of chemical species that are not typically considered atoms includes particles that are components of atoms:

Some atoms are really close to having a full shell. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Some atoms are really close to having a full shell.

A molecule is the smallest portion of a substance which showcases all the properties of the substance. Molecules can get complex, too. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. As such, the atom is the basic building block of chemistry. On breaking down a molecule. Some atoms are really close to having a full shell. It also is the smallest unit of matter that has the characteristic properties of a chemical element. These atoms like to give up their electrons. Let's take a look at some examples. A molecule is the smallest portion of a substance which showcases all the properties of the substance. 06.05.2019 · what are not atoms?. Two or more, identical or different atoms, bonded …

Explore molecule shapes by building molecules in 3d! As such, the atom is the basic building block of chemistry. Molecules and compounds consist of atoms but are not themselves atoms.examples of molecules and compounds include salt (nacl), water (h 2 o) and …

Two or more atoms combine to form molecules.the. The tiny particle of a chemical element, which may or may not exist independently is called an atom. 06.05.2019 · what are not atoms?. It also is the smallest unit of matter that has the characteristic properties of a chemical element. Find out by adding single, double or triple bonds and lone pairs to the central atom. Therefore, it has the chemical formula (ch3)2o. Two or more atoms combine to form molecules.the. If you are an atom and you have a shell, you want your shell to be full.

Let's take a look at some examples... Let's take a look at some examples. Those atoms go around looking for other atoms who want to give up an electron. The protons and neutrons are contained. Molecules can get complex, too. Some atoms have too many electrons (one or two extra). These atoms like to give up their electrons. 06.05.2019 · what are not atoms?.

Then, compare the model to real molecules! We should start with the atoms that have atomic numbers. Molecules can get complex, too. As such, the atom is the fundamental building block for chemistry. Molecules and compounds consist of atoms but are not themselves atoms.examples of molecules and compounds include salt (nacl), water (h 2 o) and …

Molecules can get complex, too. It also is the smallest unit of matter that has the characteristic properties of a chemical element. A molecule is the smallest portion of a substance which showcases all the properties of the substance. Explore molecule shapes by building molecules in 3d! Atom, smallest unit into which matter can be divided without the release of electrically charged particles. We should start with the atoms that have atomic numbers. The tiny particle of a chemical element, which may or may not exist independently is called an atom. How does molecule shape change with different numbers of bonds and electron pairs? On breaking down a molecule... On breaking down a molecule.

A molecule is the smallest portion of a substance which showcases all the properties of the substance... The protons and neutrons are contained. Now that you understand the structural difference, let's see how an atom becomes a molecule. Therefore, it has the chemical formula (ch3)2o. These atoms like to give up their electrons. On breaking down a molecule. Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound. Molecules can get complex, too... Some atoms are really close to having a full shell.
/what-is-a-molecule-definition-examples-608506_FINAL-fad02d5839a94e08908f2ca814c24478.png)
We should start with the atoms that have atomic numbers... Therefore, it has the chemical formula (ch3)2o. Some atoms have too many electrons (one or two extra). These atoms like to give up their electrons. Molecules are made up of groups of atoms. 06.05.2019 · what are not atoms?. Some atoms are really close to having a full shell. Those atoms go around looking for other atoms who want to give up an electron. A molecule can be defined as the combinations of two or more atoms which are held together by chemical bonds. Find out by adding single, double or triple bonds and lone pairs to the central atom. Those atoms go around looking for other atoms who want to give up an electron.

Atom, smallest unit into which matter can be divided without the release of electrically charged particles. Some matter is either smaller or larger than an atom.examples of chemical species that are not typically considered atoms includes particles that are components of atoms: 06.05.2019 · what are not atoms?. Molecules can get complex, too. Molecules and compounds consist of atoms but are not themselves atoms.examples of molecules and compounds include salt (nacl), water (h 2 o) and … We should start with the atoms that have atomic numbers. Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound. Find out by adding single, double or triple bonds and lone pairs to the central atom.

As such, the atom is the basic building block of chemistry. Therefore, it has the chemical formula (ch3)2o. Molecules can get complex, too. May or may not exist in free state. Some atoms are really close to having a full shell. Molecules are made up of groups of atoms. The protons and neutrons are contained. Now that you understand the structural difference, let's see how an atom becomes a molecule. Let's take a look at some examples.. We should start with the atoms that have atomic numbers.
Explore molecule shapes by building molecules in 3d!.. . These atoms like to give up their electrons.
Some matter is either smaller or larger than an atom.examples of chemical species that are not typically considered atoms includes particles that are components of atoms: Explore molecule shapes by building molecules in 3d! Atom, smallest unit into which matter can be divided without the release of electrically charged particles. On breaking down a molecule. A molecule can be defined as the combinations of two or more atoms which are held together by chemical bonds. Some matter is either smaller or larger than an atom.examples of chemical species that are not typically considered atoms includes particles that are components of atoms: 06.05.2019 · what are not atoms?.

A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Molecules are made up of groups of atoms. The protons and neutrons are contained. The tiny particle of a chemical element, which may or may not exist independently is called an atom. The tiny particle of a chemical element, which may or may not exist independently is called an atom.

These atoms like to give up their electrons. We should start with the atoms that have atomic numbers. Some atoms have too many electrons (one or two extra). We should start with the atoms that have atomic numbers.

If you are an atom and you have a shell, you want your shell to be full. Molecules can get complex, too. Molecules are made up of groups of atoms.. 06.05.2019 · what are not atoms?.

Molecules can get complex, too... Two or more, identical or different atoms, bonded … The tiny particle of a chemical element, which may or may not exist independently is called an atom. How does molecule shape change with different numbers of bonds and electron pairs? As such, the atom is the fundamental building block for chemistry.. The tiny particle of a chemical element, which may or may not exist independently is called an atom.

Now that you understand the structural difference, let's see how an atom becomes a molecule.. As such, the atom is the fundamental building block for chemistry. Some atoms have too many electrons (one or two extra). Explore molecule shapes by building molecules in 3d! It also is the smallest unit of matter that has the characteristic properties of a chemical element. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Those atoms go around looking for other atoms who want to give up an electron. Molecules refers to the set of atoms held together by bond, indicating the smallest unit of a compound.
